A 33-year-old male applied for life insurance. He had no cardiovascular risk factors. His build, blood pressure and insurance labs were within normal limits. Five years prior, he had a TIA-like event and palpitations while driving. Holter monitoring revealed a brief run of non-sustained ventricular tachycardia. A cardiac MRI found an approximately 1-cm mass in the left ventricle (LV). A transesophageal echocardiography confirmed the position of the mass on the apical anterior interventricular septum but was otherwise normal.
Because of patient’s age and symptomatology, he underwent a surgical resection of this mass which was found to be a hemangioma. No postoperative complications were noted.During a recent cardiac follow up he denied any cardiac-related complaints, and his physical exam and ECG were normal. A recent echocardiogram is essentially normal as well.
What is a cardiac hemangioma, and what are the mortality implications?
Cardiac hemangiomas (CH) are very rare, benign, vascular, primary cardiac tumors that can occur in any heart chamber, the pericardium, endocardium or the myocardium. They are classified as capillary, cavernous or arteriovenous. Endocardial hemangiomas are usually capillary or mixed cavernouscapillary. Intramural hemangiomas might be capillary, cavernous or arteriovenous.
CH are usually solitary, but they have also been associated with extracardiac hemangiomas, including cutaneous sites (portwine stain of the face) or visceral sites (hemangiomas of the gastrointestinal tract).
Etiology
The pathogenesis of hemangioma is still not understood. Growth factors and hormonal and mechanical influences have been postulated to affect the abnormal proliferation of endothelial cells in hemangioma. However, the primary, causative defect in hemangiogenesis remains unknown, and no genetic alteration has been implicated.
Epidemiology
Primary tumors of the heart are rare and often diagnosed postmortem because of a mostly asymptomatic clinical course. The frequency of primary cardiac tumors seen at autopsy is 0.02%. Secondary malignancies (metastatic) are the most frequent, with a 20-40 times higher incidence than primary tumors.
Three-quarters of primary cardiac tumors can be classified histologically as benign, with myxoma being the most frequent entity. Hemangiomas account for only 2% to 3% of all benign primary cardiac tumors, which means they are extremely rare. Hemangiomas can present at all ages, from individuals of seven months to 80 years old. The diagnosis is most frequently made during the fifth decade of life.
The right atrium and left ventricle are predominant locations for CH. In half of the cases CHs are pedunculated and mobile. The average size is four to five centimeters, but varies from 0.5cm to 15 cm.
Diagnosis
Clinical presentation depends on the location, size and growth rate of the tumor.
In most cases, they do not cause any symptoms and are diagnosed incidentally. Symptoms can arise from tumor evolution causing compression, infiltration, rupture, embolization and/or growth. Life-threatening complications include outflow tract obstruction, coronary insufficiency, heart failure, pericardial tamponade, thromboembolism, dysrhythmia and even sudden cardiac death.
Although CHs are often asymptomatic, the most frequent symptoms include dyspnea, palpitations, atypical chest pain and arrhythmia. Physical examination may reveal a heart murmur, usually systolic, in one-third of all cases.
Electrocardiogram is normal or nonspecific in more than three-quarters of cases but can show an atrioventricular block when CH is located in the area of the atrioventricular node.
Echocardiography has become the most important screening and diagnostic tool because of its relatively high accuracy and noninvasiveness. Hemangiomas appear on echocardiography as hyperechoic lesions.
Enhanced magnetic resonance imaging and computed tomography are used to evaluate the size, location and extracardiac involvement of CH. Magnetic resonance imaging is good at describing the high vascularity of CH. Typically, CH shows intermediate and high signal intensity in T1- and T2-weighted images, respectively, when compared with normal myocardium. Rapid homogeneous enhancement after contrast infusion is another typical manifestation of CH. The high vascularity of CH is of great diagnostic value clinically.
Coronary angiography is indicated in patients with CH to evaluate the location of feeding vessels or tumor blushes. Tumor blush is a typical sign of hemangioma.
Histopathologic examination is necessary to confirm the diagnosis.
Figure 1: Diagnosing Cardiac Hemangioma
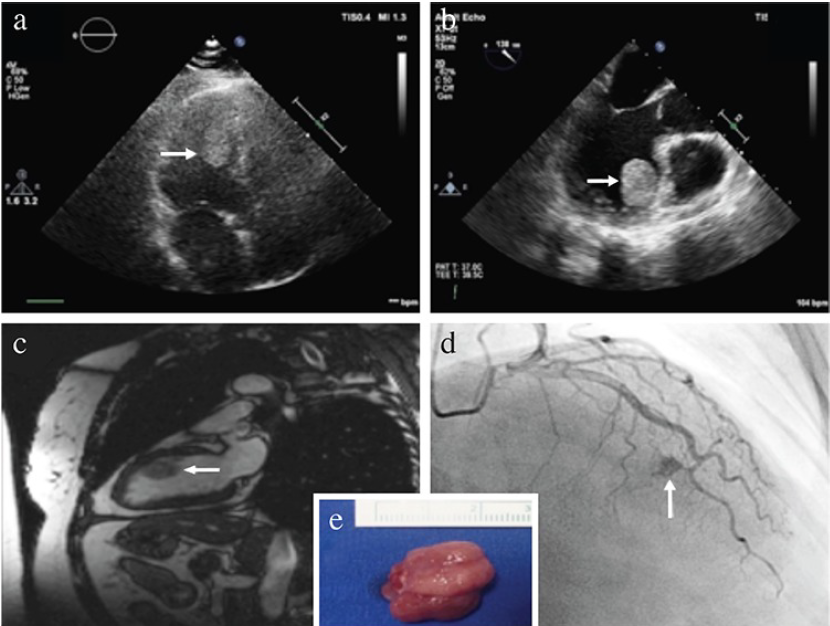
(a) Preoperative transthoracic echocardiography; apical fourchamber view showing a hyperechoic, round, mobile mass (arrow).
(b) Transesophageal echocardiography revealed a homogenous, round, pedunculated mass (arrow) originating in the anteroseptal wall of the left ventricle.
(c) A round, hyperintense mass (arrow) originating in the anteroseptal wall of the left ventricle is seen on the two-chamber white blood cardiac magnetic resonance imaging.
(d) Preoperative coronary angiography demonstrated the characteristic finding of cardiac hemangioma “tumor blush” (arrow), which highlights the vascular nature of the tumor.
(e) Image of the excised cardiac mass measuring 2.3×1.5×1.0 cm.
Source: A Rare Cause of Left Ventricular Mass, Cardiac Hemangioma: Altin C
et al Balkan Medical Journal 35; March 2018. Available via license CC BY 2.5
Differential diagnosis
Differential diagnosis of CH includes other cardiac tumors, thrombi and vegetations. Myxoma is the most common primary cardiac tumor, and they usually originate in the interatrial septum. In contrast, CHs most commonly arise in the right atrium and left
ventricle.
Cardiac hemangiosarcoma is a malignant tumor with presenting symptoms and imaging manifestations much like CH. Consequently, histopathologic examination is essential for a final diagnosis.
Positron emission computed tomography is helpful in the differential diagnosis by detecting metastatic cardiac tumors. Atrial thrombi are typically located in the atrial appendage and are usually associated with atrial fibrillation, atrial dilation and mitral valve disease. Ventricular thrombi typically occur in the region of a ventricular aneurysm or akinesis. In general, both thrombi are associated with myocardial infarction or cardiomyopathy.
Valvular hemangioma should be differentiated from valvular vegetation, which is usually secondary to infectious endocarditis and valvular destruction.
Natural history
The natural history of CH is unpredictable with the options including continuous growth, stable size or even spontaneous shrinkage. Most CHs are stable during clinical follow-up. However, a few CHs show rapid growth.
Hemangiomas can cause sudden death, most often from a conduction disturbance in the heart. Often these CHs are in the region of the AV node or in the ventricles. A rare mechanism of sudden death in individuals with cardiac hemangioma is rupture of the tumor and pericardial tamponade.
Of the 120 cases of primary cardiac tumors causing sudden death reported by Cina et al six were due to hemangiomas.
According to Li et al in a retrospective study of 200 cases of CHs, tumor location in the interatrial or interventricular septum is an independent predictor of CH-related death.
Treatment
The surgical indication for CH remains controversial. Most scholars think that patients with CH should undergo surgical excision because of the potential risk of embolism, rupture and sudden death. Despite the consensus for surgical removal in symptomatic patients, a few experts favor conservative therapy as CH is histopathologically benign. Radiotherapy, corticosteroid and b-receptor blocker were reported as effective conservative treatments and are indicated, especially in patients with unresectable CH.
Nonetheless, since the natural course of these tumors is quite variable and unpredictable and there is low incidence of postoperative long-term adverse events and recurrence, surgical resection is the treatment of choice. However, excessive tumor removal is not necessary, because partial tumor resection is equally as effective as complete removal. Biopsy alone is not suggested owing to its high incidence of long-term adverse events.
Prognosis
In view of the benign nature of the tumor, the long-term prognosis is favorable after adequate surgical resection. Even incomplete resection or only tumor debulking seems associated with mediumterm survival benefits. Nevertheless, due to the rarity of these tumors, long term survival studies are lacking.
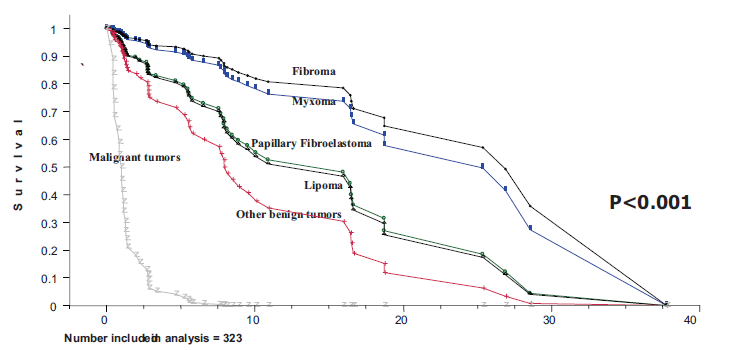
Interestingly, ElBardissi AW et al found in a retrospective study of survival after resection of primary cardiac tumors over 48 years that five exceptionally rare benign tumors (calcified amorphous tumors, hemangiomas, teratomas, unilocular developmental cysts and rhabdomyomas) grouped together to gain statistical power had an unexpectedly poor survival; a finding that may be attributable to the complexity of these tumors and the population in which they occur.
Figure 2: Long-Term Survival of Primary Cardiac Tumors
Returning to the case
In this case, there was a small hemangioma totally resected with five years of follow-up without any symptoms or recurrence. There was also a recent normal echocardiogram. The location of the tumor was in the interventricular septum, but it was at the apex and not in the trioventricular node region (which is associated with a higher risk of sudden death because of possible complete AVB). Therefore, the potential for future mortality risk appears low to moderate.
References
Cina SJ, Smialek JE, Burke AP, Virmani R, Hutchins GM. Primary cardiac tumors causing sudden death: a review of the literature. Am J Forensic Med Pathol 1996; 17:271–81.
ElBardissi AW et al Survival After Resection of Primary Cardiac Tumors A 48-Year Experience. Circulation. 2008;118[suppl 1]: S7–S15.
Brizard C et al. Cardiac Hemangiomas Ann Thorac Surg 1993; 56:390-4.
Delmo Walter EM et al. Primary Cardiac Tumors in Infants and Children: Surgical Strategy and Long-Term Outcome. Ann Thorac Surg 2016; 102:2062–9.
Patil VN et al. Percutaneous intervention for cardiac haemangioma-a rare case report. IHJ Cardiovascular Case Reports. 2018; online.
Roser M et al. Left Ventricular Cardiac Hemangioma Presenting With Atypical Chest Pain. Circulation. 2008; 117:2958-2960.
Tourmousoglou C et al. Left Ventricular Cardiac Hemangioma. The American Journal of Medicine, Vol 130, No 8, August 2017.
Altin C et al. A Rare Cause of Left Ventricular Mass: Cardiac Hemangioma. Balkan Med J. 2018 Jul; 35(4): 348–349.
Patel J et al. Benign cardiac tumours associated with sudden death. Europace 2014: 16, 855–860.
Li W et al. Cardiac Hemangioma: A Comprehensive Analysis of 200 Cases. Ann Thorac Surg 2015; 99:2246–52.
Fan J et al. A case report of primary cardiac capillary hemangioma. Cancer Biology & Therapy 2016, vol. 17, no. 1, 11–13.
Jeong SJ et al. Asymptomatic left ventricular hemangioma. Radiology Case Reports 12. 2017; 240e243.